Consensus statement of the Swiss Society of Sports Medicine
Clénin GE1, Cordes M1, Huber A2, Schumacher YO3, Noack P4, Scales J5, Kriemler S6
1 Sportmedizinisches Zentrum Ittigen bei Bern, Haus des Sports, 3063 Ittigen, Switzerland; german.clenin@smzbi.ch
2 Zentrum für Labormedizin, Kantonsspital Aarau, Tellstrasse, 5001 Aarau, Switzerland
3 Aspetar Orthopedic and Sports Medicine Hospital, Doha, Quatar
4 Zentrum für Medizin und Sport im Säntispark, 9030 Abtwil, Switzerland
5 Bannockburn Health Centre, Stirling, Scotland
6 Epidemiology, Biostatistics and Prevention Institute, University of Zürich, Switzerland
This article was first published in October 2015 in Swiss Medical Weekly (SMW).
We kindly thank the Board of editors of SMW to allow the re-publication of this entire consensus paper in our journal!
Abstract
Iron deficiency is frequent among athletes. All types of iron deficiency may affect physical performance and should be treated. The main mechanisms by which sport leads to iron deficiency are an increased iron demand, an elevated iron loss and a blockage of iron absorption due to hepcidin bursts. As a baseline set of blood tests, haemoglobin, haematocrit, mean cellular volume (MCV), mean cellular haemoglobin (MCH) and serum ferritin levels are the important parameters to monitor iron deficiency. In healthy male and female athletes >15 years, ferritin values <15µg/l are equivalent to empty, values from 15 to 30µg/l to low iron stores. Therefore a cut-off of 30µg/l is appropriate. For children aged from 6–12 years and younger adolescents from 12–15 years, cut-offs of 15 and 20µg/l, respectively are recommended. As an exception in adult elite sports, a ferritin value of 50µg/l should be attained in athletes prior to altitude training, as iron demands in these situations are increased.
Treatment of iron deficiency consists of nutritional counselling and oral iron supplementation or, in specific cases, by intravenous injection. Athletes with repeatedly low ferritin values benefit from an intermittent oral substitution. It is important to follow up the athletes on an individual basis with the baseline blood tests listed above twice a year. A long-term daily oral iron intake or iv-supplementation in the presence of normal or even high ferritin values does not make sense and may be harmful.
Zusammenfassung
Eisenmangel im Sport ist häufig und relevant. Alle Stadien des Eisenmangels beeinflussen die körperliche Leistungsfähigkeit und sollten deshalb behandelt werden. Die Hauptmechanismen, welche bei Athletinnen und Athleten zu einem Eisenmangel führen, sind: ein erhöhter Eisenbedarf, ein vermehrter gastrointestinaler Eisenverlust und eine Blockierung der Eisenabsorption aufgrund erhöhter Hepcidinwerte.
Als Basisblutentnahme sollen Hämoglobin, Hämatokrit, MCV (mittleres zelluläres Volumen), MCH (mittleres zelluläres Hämoglobin), C-reaktives Protein und Ferritin bestimmt werden. Bei gesunden Sportlerinnen und Sportlern entspricht ein Ferritinwert von <15µg/l leeren und ein solcher von 15–30µg/l niedrigen Eisenspeichern. Aus diesem Grund ist ein Ferritin-Cutoff von 30µg/l für die Diagnosestellung des Eisenmangels sinnvoll. Für Kinder von 6–12 Jahren und Jugendliche von 12–15 Jahren beträgt der empfohlene Cut-off Wert 15 bzw. 20µg/l. Als Ausnahme soll im Leistungssport bei Erwachsenen vor Antritt eines Höhentrainingslagers ein Ferritinwert von 50µg/l erreicht werden, da der Eisenbedarf in dieser Situation deutlich erhöht ist.
Die Behandlung des Eisenmangels besteht aus einer Ernährungsberatung und oraler Therapie oder, in spezifischen Fällen, aus einer intravenösen Eisengabe. Athletinnen und Athleten mit wiederholt niedrigen Ferritinwerten profitieren von einer oralen Erhaltungstherapie. Es ist wichtig, die Sportlerinnen und Sportler mit der Basisblutentnahme zweimal jährlich individuell zu begleiten. Eine Langzeittherapie mit Eisen peroral oder eine intravenöse Eisengabe bei normalen oder sogar hohen Ferritinwerten ist nicht sinnvoll und wahrscheinlich schädlich.
Introduction
Iron deficiency among athletes, in males and more often in females, is a commonly encountered condition for the sports medicine physician. Iron deficiency is one of the most common deficits globally with a clear predominance in adolescence and in menstruating females [1]. Data from a general Swiss population show frequencies for iron deficiency for menstruating females of 22.7%, for male military recruits of 7.2% and for iron deficiency anaemia of 2.2% (females) and 0.1% (males) [2,3]. In sports the rate of iron deficiency is distinctly higher up to 52% in female adolescent athletes [4,5,6] and occurs more often in endurance sports and in disciplines with a high prevalence of eating disorders. These abnormal findings need a careful clinical look as iron deficiency affects many organ systems of the body and not just oxygen transport, especially in sports [7].
On the other hand, Switzerland has recently experienced some kind of “iron hype” for different reasons. Not all of them seem to be rational [8,9]. The problem is enhanced by the finding of an earlier study among Swiss top athletes, showing, that iron supplements were consumed to some extent uncritically and in excess. [10,11]
Sports medicine physicians are often in charge of male and female athletes at their peak performance. In this role, they are aware of the importance of adequately diagnosing and treating iron deficiency in all its aspects, but at the same time to prevent iron overload. This article is a consensus statement of the Swiss Society of Sports Medicine and provides an overview and practical guidelines for the diagnosis and treatment of iron deficiency in sports and should help clinicians in decision making.
Definition of iron deficiency
and general function of iron
Iron is a transition metal and has multiple functions in more than 180 biochemical reactions in the human body including electron transport in redox reactions (cytochromes, sulphuric proteins), redox catalytic functions (cytochrome p450, catalase, peroxidase) and reversible storage and transport of O2 (haemoglobin, myoglobin). It also plays an important role in the production of neurotransmitters, and is essential in synaptogenesis and myelinisation. Moreover oxidative phosphorylation is the most critical biochemical pathway in which iron is involved [8,12].
Figure 1 gives an overview of iron distribution in the human body. The total body content of iron amounts to approximately 4g in men and 2.5g in women. This iron is divided up between 3 active sites, firstly haemoglobin, myoglobin and enzymes. The rest (20% of total) remains as inactive, depot iron in the form of ferritin and hemosiderin. Finally 0.2% of the total iron exists as transport iron in the form of transferrin. In adolescents the relative amount of iron in the different compartments is comparable but may vary slightly depending on body size and initiation of menses (fig. 1) [8].
The usual loss of iron (1mg per day in males and 2mg per day in females) due to gastrointestinal epithelial shedding and menstruation is compensated by absorption in the small intestine [13,14]. Of the ingested iron of 10–14mg, the enterocytes absorb only about 0.5–2mg (5–15%) [14,15]. Nevertheless, during increased losses (e.g. microischemia in sports, bleeding, haemolysis) and elevated demand (e.g. growth with the building up of the adult haemoglobin mass, pregnancy), adequate uptake is guaranteed through an up to four-fold elevated intestinal absorption as long as sufficient iron is provided by nutritional intake [14].

The absorbed iron is stored in ferritin in the cytoplasm of the enterocytes [12]. To export the iron to the plasma the iron is carried out by ferroportin on the basolateral surface of the enterocytes. There, iron is bound to transferrin and transported to the liver where it is stored as ferritin or transferred to the iron consuming tissues e.g. bone marrow.
Ferroportin is important in the tight regulation of iron homeostasis. The main regulatory constituent is hepcidin [16]. Synthesized in hepatocytes, hepcidin regulates the iron export out of the storing cells. In phases of high iron loading and in response to inflammatory processes the synthesis of hepcidin is increased leading to the internalisation of ferroportin on enterocytes which in turn blocks iron transportation into the circulation. The same mechanism leads to a blockade of iron within the macrophage system, thus preventing the transfer of iron from macrophages to erythroblasts, the precursors of erythrocytes [12] (fig. 2).
Any type of exercise will also cause some kind of inflammation in the body, as this inflammation and the subsequent repair mechanisms are the basis of adaptation to training. The size of the inflammatory response depends on the type, intensity and duration of the training. Several markers of iron metabolism are affected by the inflammatory cascade, as they are part of the acute-phase response [18]. Concerning iron metabolism, intensive training has been shown to lead to distinct increases in hepcidin [5,19,20].

This leads to a block of iron absorption, disruption of iron transfer from macrophages to erythroblasts and may possibly induce iron deficiency. Hepcidin synthesis is, on the other hand, suppressed by erythropoetic activity and anaemia. This allows an increased intestinal absorption and utilization of iron from the macrophages and hepatocytes under conditions of elevated iron loss or increased demand [21,22].
Up to now the main mechanism by which sport causes an increase of iron loss was explained by microischemia of the gut during excessive training [23,24]. Losses through excessive sweating [25] and possible blood loss in the urinary tract [26] are in absolute terms not relevant. The foot-strike haemolysis which describes the mechanical destruction of red blood cells in the foot of the running athlete does exist [27,28], but as the salvage of blood constituents is complete there is no loss of iron. Newer results show that there is the additional mechanism explained above where participation in intensive sport triggers hepcidin bursts, causing blockage of iron [20].
Figure 3 describes the different stages of progressive iron deficiency: When iron losses exceed absorption or absorption falls below demand, initially iron stores will deplete, resulting in a reduced ferritin level. At a certain point, the stored iron is too low to provide the tissues with sufficient iron. This will induce the production of zinc-protoporphyrin (ZnPP) and an increase of soluble transferrin receptor (sTfR).

As haemoglobin, mean cellular volume (MCV), and mean cellular haemoglobin (MCH) are still normal, this condition is called non anaemic iron deficiency (NAID). NAID is defined as a deficiency of iron without affecting haematopoiesis.
If iron balance remains negative, the youngest red cells will be insufficiently haemoglobinised and thus appear as hypochromic and microcytic with lower MCH and MCV than the entire cell population. If progression of iron deficiency continues, MCH and MCV will drop below the lower limit of the normal range (28pg and 80fl, respectively) and iron deficiency with microcytosis and/or hypochromia (IDMH) develops. IDMH is defined as an iron deficiency affecting haematopoiesis. In this case, ferritin is <30µg/l, the red cell indices are mostly but not always affected and the concentration of haemoglobin is still normal (men >140g/l, women >120g/l) [29].
Ultimately, haemoglobin concentrations will drop below the lower limit of the normal range and frank iron deficient anaemia (IDA) is established [29]. In IDA ferritin and haemoglobin are lowered and the red cell indices are reduced or normal [29].
A further particular situation is the functional iron deficiency encountered in patients with anaemia of chronic disorder (ACD), or tumor or haemodialysis patients. The iron demand is higher than the iron supply out of the iron stores. In consequence hypochromic reticulocytes and erythrocytes are formed, the haemoglobin in reticulocytes (CHr respectively Ret-He, table 1) falls < 28pg [30,31]. This situation is a result of elevated Interleukin 6 and hepcidin levels seriously impairing iron turnover and may therefore occur with normal or even elevated ferritin values, reflecting normal iron stores. [21,31]
The basic measurements are shown in figure 4: these are haemoglobin, haematocrit, and erythrocyte count, with calculation or measurement of the red cell indices MCV and MCH. Table 1 gives an overview and explains cut off values of relevant tests to diagnose NAID, IDMH and IDA.

Ferritin is the most widely used parameter in the evaluation of iron deficiency. Since ferritin also acts as an acute phase protein each inflammatory process should be excluded by the patient history. Additionally, it should be considered that even heavy/powerful sporting activities might increase acute phase reactants [40–42,37]. Depending on duration and intensity of the activity ferritin values may stay normal, show a rise of 27% [40] with a return to baseline within a day or in case of ultramarathons be double as high as the prerace value and return to baseline only after 6 days [42,43].
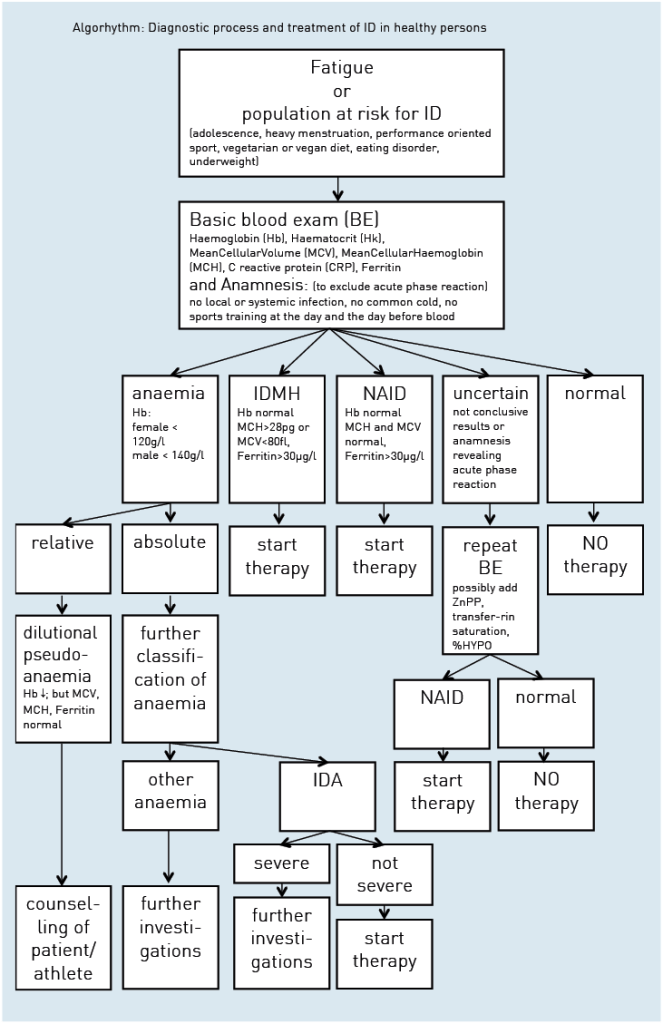
Free serum iron has a high daytime and a high variability between one person and another. Morning values are at a peak more than twice as high as values measured 12 hours later, so it cannot be used to represent iron in the body. Furthermore, free serum iron is lowered with acute phase reactions and elevated in cases of haemolysis after blood sampling. Nowadays it is an obsolete marker and should only be used to calculate transferrin saturation or in situations of acute iron intoxication [29]. Transferrin saturation has some benefit for classification of iron deficiency. A cut off of 20% and below is accepted to define iron deficiency [29]. As free serum iron is needed for its calculation, one must be aware of this limitation, particularly in inflammation where transferrin saturation may be between 10 and 20% without iron deficiency.
The indirect markers ZnPP [38,39] and sTfR [29,37] may be additionally useful to define IDMH and differentiate NAID from a functional iron deficiency. The sTfR was thought to be an excellent parameter because it is not influenced by inflammation and exercise. But it is mostly elevated during increased erythropoesis, so it may be of only additional value. ZnPP is reentering routine testing now and gives additional information to judge iron status of erythropoesis.
Hepcidin has been established as a key regulator of iron absorption from the enterocytes and release from the macrophages; however, it has not entered routine testing [16,21].

Iron deficiency in sports –
influence on performance
Dilutional pseudoanaemia
Anaemia is defined by a lowered haemoglobin concentration in the venous blood sample. This definition neglects that in fact anaemia is the real reduction of the total haemoglobin mass in the body, a so-called absolute anaemia (see fig. 4). In contrast, relative or dilutional pseudoanaemia is defined as a lowered haemoglobin and haematocrit owing to a distinctly elevated plasma volume with normal red cell mass and total haemoglobin mass. The other blood parameters, particularly ferritin, MCV, and MCH are within the normal range and performance is not affected. This plasma volume increase is considerable and is due to repeated bouts of physical training over the years. Its magnitude depends on the intensity and duration of the effort. In about 10–15% of mainly endurance athletes, especially when training time exceeds 10 hours per week, this dilutional pseudoanaemia can be observed [44].
The same dilutional effect is found in ultralong endurance disciplines or sports events lasting several days (e.g. road cycling tours). As seen in figure 5 in male and female athletes with dilutional pseudoanaemia the increase in plasma volume causes a slight lowering of the haematocrit while the red cell mass stays high [42,45–48].
This is why top level endurance athletes show a high correlation between haemoglobin mass and endurance capacity, but a low correlation between haematocrit or haemoglobin concentration and endurance capacity [49,50]. Therefore, the haemoglobin mass is the important factor for endurance capacity, while having a high plasma volume accompanied by a low haematocrit is by no means a disadvantage [51–53]. A larger intravascular volume has beneficial effects on performance, as it improves thermoregulation, increases cardiac pre-load and enhances the body’s ability to dilute metabolites. The exact mechanism for this adaptation remains to be explored, and shifts in proteins and electrolytes are thought to play a role [54]. Exercise induced plasma volume shifts are usually reversible within days after cessation of the physical effort [40].
A dilutional pseudoanemia cannot be ruled out by a venous blood exam only, but in clinical practice serial measurements of the basic blood exam are helpful. These include measurements during the training pause at the end of the season (hematocrit, Hk↑), during the season (Hk↓), once immediately after an intensive and long lasting training session (Hk↑) and 24–48h after this session (Hk↓). If these serial measurements do not reveal the expected variations of the hematocrit (showing at least normal values in the training pause at season’s end) the measurement of hemoglobin mass to confirm the dilutional pseudoanaemia may be considered with, for example, the CO rebreathing method [48–50].
Excessive haematocrits
Unfortunately, top level road cyclists fifteen years ago often showed excessive haematocrit levels over 50% exceptionally up to 60%. Based on confessions of several riders, these values could only be reached by mostly systematic doping procedures with mainly erythropoietin, growth hormone and testosterone. During longer lasting competitions blood transfusions (re-transfusion of own blood, collected several months before) [55] were given to boost oxygen carrying capacity. Through the awareness of these doping procedures, health control tests with blood sampling with upper limits of haematocrit (e.g. the 50% rule of the Union de Cyclisme International [56]) or haemoglobin (e.g. 170g/l for male, 160g/l for female of the Fédération International de Ski [57]) to protect athletes from fatal thrombotic events were introduced. These were followed by newer doping tests (e.g. EPO-tests, growth hormone) and antidoping strategies like the biological passport [58]. Several blood samples collected over the year provide an individual blood profile and allow individual reference values to be established for each athlete [59]. This biological passport proposed by the WADA (World antidoping agency) gives a much closer follow-up and is used in cases of violation of the antidoping code [58].
Influence of iron on physical performance
Iron is in addition to its function in oxygen transport a key component of the enzymatic system of the respiratory chain. These presumably distinct roles were investigated in early animal studies [60,61,62]. In these studies, the authors aimed at differentiating between a decline in performance due to anaemia and a decline due to enzymatic impairment. Using an animal model in a cross over setting with iron depletion and cross over transfusion to correct for anaemia, it was shown that not only anaemia but also iron depletion without anaemia led to a significantly decreased number of mitochondria and reduced activity of the respiratory enzymes. The authors therefore postulated that iron depletion without anaemia affected oxidative capacity, whereas anaemia affected mostly oxygen transport [60,61,62].
At present the cut-off of ferritin is still debated with values ranging from 15µg/l by WHO [73] to 16–32µg/l [74,75] in studies that used the “gold standard procedure of bone marrow staining”. It has to be noted that studies investigating this matter were not all conclusive and some presented controversial results [76,77].
If not only erythropoiesis but also clinical symptoms of an iron deficiency such as fatigue (but not yet performance) are considered, the cut-off may be slightly higher. Recently Krayenbuehl et al. showed in their double blind randomised study with intravenous iron administration in nonanaemic premenopausal women with low ferritin and fatigue an improvement in mood state in the ferritin group <15µg/l [78]. Earlier, Verdon et al analysed fatigue and mood state with oral therapy in a family doctor’s office setting. They found that non anaemic women with ferritin concentrations below 50µg/l improved with oral supplementation [79]. Vaucher et al came to comparable results [80]. But their proposed cut-off of 50µg/l must be interpreted with caution because of methodological concerns (e.g. assessment of stool coloration by iron not blinded, definition of iron deficiency based on a limited number of variables).
A few studies have integrated a performance measure (table 2). McClung et al showed in female military recruits with oral iron treatment versus placebo an improvement in running performance in recruits with IDA. Those with NAID showed an improved mood state, but no change in performance. Since then, several other studies have shown a performance impairment in non anaemic iron depleted endurance athletes [68–70,81,72]. A limiting factor of all these studies is the influence of training or mild changes in haematological parameters of other origin, both of which are difficult to quantify. Garvican et al performed very recently a study with 27 highly trained distance runners (13 male and 14 female athletes) with measurement of haemoglobin mass and endurance performance. They compared in a randomized controlled setting intravenous iron versus six weeks of oral treatment with one group having clearly iron deficient stores (ferritin <15µg/l or ferritin <35µg/l and transferrin saturation <20%) and the other with better iron stores but a ferritin level of <65µg/l. Both forms of supplementation substantially increased ferritin levels, but haemoglobin concentration did not change in any group. Haemoglobin mass increased in the iron deficiency group with intravenous treatment accompanied by an increase in VO2max and run time to exhaustion whereas the group with ferritin <65µg/l did not show any changes in these parameters [53,64].
As multiple muscle enzymes are affected other functions related to performance have been investigated: Brutsaert et al found an increase in knee extensor strength after iron supplementation in nonanaemic iron depleted volunteers using a randomized controlled study design, which points towards a role of iron in resistance to fatigue and adaptation to training [82]. Very recently Della Valle et al investigated female rowers with NAID under oral iron supplementation for six weeks in a double blind design. They were able to show an improvement of the iron status and a better energetic efficiency in the iron group [65].
Based on the cited results and according to several review articles a ferritin cut-off of 30µg/l in adults seems to be most plausible. [8,83–90]

Iron deficiency in Sports – Therapy
Nutrition
The first step in therapy of iron deficiency is the correction of the nutritional iron intake. In nutrition, haem iron (mainly in meat) and free iron as Fe2+ or Fe3+ exist. Oral uptake studies show that the uptake of haem iron is much better than the uptake of free iron [91]. For the latter, uptake of Fe2+ is better than uptake of Fe3+ [92]. Meat, liver, poultry or fish contain haem iron as well as free iron. A vegetarian diet contains only free iron. The bioavailability for iron is very variable and depends much on actual iron stores [93] ranging from 5% to 15%. In the case of iron deficiency a significant increase in iron bioavailability up to 35% can be observed [94]. Furthermore, iron uptake in the intestinal tract is influenced by different nutritional factors including enhancers and inhibitors. Substances enhancing iron uptake are vitamin C, peptides from partially digested muscle tissue, fermented food, organic acids like malate or citrate. Substances inhibiting iron uptake are phytates, oxalates, polyphenols (black tea and coffee), peptides from partially digested vegetable proteins and calcium [83,84]. The nutritional intake should be 14mg per day. General recommendations for an optimal dietary iron intake in sports include an adequate energy intake especially for athletes with low body mass index as they suffer more frequently from iron deficiency [95]. Whether catabolism related to low energy intake influences the hepcidin regulation and down-regulates iron uptake remains open to debate. In general, regular consumption of meat, poultry or fish, at least 5 times per week is recommended as it is the main contributor of nutritional iron intake. Complementary eating of wholemeal products and daily legumes and green vegetables is suggested. Furthermore, it is beneficial to replace tea and coffee by a glass of orange or citrus fruit juice with an iron containing meal as vitamin C enhances iron uptake [84,94]. For vegetarians the goal is to reach a high load of iron through their vegetable diet. Even if nutrition is important in iron homeostasis in the human organism an IDA cannot be corrected by nutrition alone, as this would mean to eat kilograms of iron containing products (e.g. liver).
Oral iron
Usually dietary counselling and oral iron therapy are combined. Oral preparations differ in their amount and type of iron (Fe2+ or Fe3+), their complex forming substrate and in their galenic form. Novel products combine iron with vitamin C. In a dosage finding study in elderly patients with IDA Rimon et al compared three dosages of oral iron: 15mg, 50mg and 150mg. They were able to show that iron supplementation at a level of the Recommended Daily Allowances (RDA,15mg of elementary iron) already led to significant increases of the iron status. In these anaemic elderly patients the dosages of 50mg, respectively 150mg of elementary iron did not show further benefit, but had significantly more side effects particularly in the highest dose group [96]. As there is further evidence that oral iron loading increases circulating hepcidin [97], the recommended dosage of oral iron shouldn’t be chosen too high. In a recent comparison of oral iron supplementation in a randomised controlled trial looking at iron status and performance in active women, 100mg of FeSO4 (approximately 20mg of elementary iron) was shown to be effective [98]. We therefore recommend a supplementation of 40 to 60mg of elementary iron once daily. Oral iron is in general well tolerated and efficient [99]. Side effects of oral therapy are mainly gastrointestinal including nausea, dyspepsia, constipation or diarrhoea [83,84,100]. They are usually not severe. Some individuals with an existing tendency to constipation benefit from drinking additional fruit juice to prevent heavy constipation. Otherwise compliance will be seriously affected.
Intravenous iron
When oral therapy fails or immediate restoration is needed iv-therapy should be considered. At the moment two different preparations are available in Switzerland including iron saccharose-complex and ferric carboxymaltose-complex. The dosage is dependent on the severity of the iron deficiency. In one application usually 200mg Fe-saccharose, 500 to 1000mg Fe-carboxymaltose can be administered.
The main advantage of the i.v.-therapy is the immediate correction of the iron deficiency and restoration of the empty iron stores. Generally, compliance with i.v. iron supplements is good. Side effects may encompass the following symptoms: a transient disturbance of taste, headache, dizziness, myalgia, fever but also severe adverse reactions such as hypotonic and anaphylactoid reactions, tachycardia and arrhythmia, dyspnoea and bronchospasm might be observed, albeit very rarely [100]. Moreover, transient and usually asymptomatic hypophosphatemia is frequently observed after the administration of Fe-carboxymaltose.
It is still under discussion whether hypophosphataemia may possibly be a cofactor for cardiac events [101]. Nowadays severe side effects are rare. For Fe-saccharose and Fe-carboxymaltose there is no fatal outcome reported in Switzerland. Internationally there is just one case with an already severely ill patient reported who had a treatment with Fe-carboxymaltose. This is in contrast to the previously used iron-dextran products with a much higher rate of severe and fatal side effects. In 2013, another type of i.v.-iron, Fe-oxytol, was withdrawn due to severe hypersensitivity reactions in four patients, including one fatal outcome, observed nine months after approval by the regulatory authorities in Switzerland [102,103]. Even with the use of the new preparations, severe adverse reactions cannot be excluded, and the administration of i.v. iron preparations is only recommended in settings where resuscitation skills are available and observation of the patient for 30 minutes after the end of the i.v. administration can be guaranteed. [104,105].
In general, the frequency of side effects seems to be lower when the i.v. iron is applied by infusion instead of slow bolus injection. Importantly, the exact dilution given by the manufacturer needs to be respected. Further, the physician needs to respect the WADA antidoping regulations concerning administration of infusion in elite sports: “Intravenous infusions and/or injections of more than 50 mL per 6 hour period are prohibited except for those legitimately received in the course of hospital admissions or clinical investigations”. [106]
Control of adequate iron stores
In sports a regular control of blood parameters is necessary, especially in endurance athletes. To monitor efficacy of therapeutic measures we recommend repeating the basic blood tests 6 to 8 weeks after the start of the nutritional measures, oral therapy or i.v.-iron administration.
Depending on the blood results therapy will be continued or modified with the aim of reaching and keeping the iron stores in the regular range. Treatment approaches may be combined and athletes with repeatedly low iron stores may besides the nutritional counselling benefit from an intermittent oral substitution to preserve iron stores, e.g. substitution with 14 or 28mg elemental iron per day or 40–60mg elemental iron two to three times per week. In vegetarian athletes a similar therapeutic approach to prevent iron deficiency is recommended: three times 40 to 60mg elementary iron per week instead of meat intake or a daily supplementation with at least 14 or 28mg/day of iron will usually cover iron demand.
As iron homeostasis is exclusively and meticulously controlled by the iron uptake through the intestinal tract [107] and as there is no pathway to eliminate iron in cases of overload, iron supplementation has to be done always carefully. In extreme cases, the chronic iron overload may lead to secondary haemochromatosis.
Furthermore, excessive supplementation of oral or i.v. iron is thought to increase oxidative stress and production of free radicals [108,109] and oxidative stress is suggested to play a role in cancer genesis [110,111]. This must be critically appraised as acute exercise leads to oxidative stress [112,113].
It has been shown in mice that oral iron supplementation enhances colonic tumor development [114]. Data in humans suggest that iron may increase the risk for colorectal cancer [115]. A recent meta-analysis showed on the one hand a tendency toward a positive association between high intake of haem iron and cancer risk. On the other hand high levels of biomarkers of iron stores implied a lower cancer risk [116]. Further prospective and experimental studies are needed to evaluate the possible influence of iron in carcinogenesis.
Iron deficiency in the child and adolescent athlete
A British study reported an iron deficiency prevalence of 21% in adolescents between 11–18 years [117,118]. Although likely to be higher in the athletic population, data about young athletes concerning iron deficiency are sparse [119,120,121].

Total iron requirements in children and adolescents are distinctly increased because of additional iron needs for the expansion of the total blood volume, mean haemoglobin mass as well as for the enhancement in lean body mass during growth [122]. In adolescent females the onset of menarche is associated with an increase of iron requirement. The mean total iron requirement for adolescents reaches 1.8mg/d for boys and 2.2mg/d for girls (in females with heavy periods it is considerably more) which corresponds to more than the double that of the preadolescent period [123,124]. Otherwise risk factors for iron deficiency, both in younger and older athletes, are the same [20,23,24,125].
Haematological norm values in children and adolescents are different from adults and should always be considered (see table 3). We recommend to define the lower level of normal ferritin as 15µg/l for children between 6–12 years, 20µg/l between 12–15 years and 30µg/l for 15–18 year old adolescents, respectively [125].
As in adults in a case of an NAID, dietary counselling is the first step often combined with oral therapy. Iron requirements (RDA) are 8mg/d for 9–13 year-old children and 11mg/d for male and 15mg/d for female adolescents older than 13 years. Careful management especially of the menstruating teenage girl and the vegetarian athlete is warranted [127]. If the iron deficiency results in IDMH or IDA, further supplementation should be considered. Comparable to the therapy in adults, oral substitution can be done with either Fe2+ or Fe3+ preparations. The dosage for Fe-preparations is 5mg/kg/d for 3 months in two or three daily doses. The Fe2+ preparation is recommended as medication of choice because ofbetter bioavailability. As in adult athletes measurement of haemoglobin, red cell indices, ferritin and the CRP after 6–8 weeks of treatment is necessary in order to observe responsiveness and compliance with the treatment.
Conclusions and recommendations
Iron deficiency in sports is frequent and relevant as all stages of iron deficiency IDA, IDMH and NAID affect physical performance. To diagnose iron deficiency haemoglobin, haematocrit, MCV, MCH and ferritin are first line parameters to assess. For a valid interpretation of the results it is necessary to exclude acute phase reactions that may interfere such as training sessions and infectious diseases (patient history and measurement of CRP). In unclear situations a second measurement of the same parameters or the additional measurement of zinc-protoporphyrin, soluble transferrin receptor and transferrin saturation may be helpful. In high level endurance athletes basic blood examinations should be made two to three times a year.
Ferritin values below <15µg/l are very specific for empty iron stores. Ferritin values from 15 to 30µg/l correspond to low iron stores. A ferritin value of 30µg/l should be taken as a reasonable cut-off for adult men and women and older adolescents (15 years and older). For younger adolescents from 12 to 15 years a cut-off of 20µg/l and for children from 6 to 12 years a cut-off of 15µg/l is recommended. In adult elite sports altitude training represents a special situation with an increased need for iron. In this case a ferritin value of 50µg/l prior to altitude training should be aimed for. Every case of severe iron deficiency warrants an extended work up, and this is also true in sports.
Therapy of iron deficiency in sports consists of the nutritional counselling including a sufficient energy intake and 5 times/week haem iron intake (meat, poultry, fish) with the addition of legumes and green vegetables (e.g. spinach, fennel) usually combined with oral iron supplementation. Daily oral iron preparations in a dosage of 40 to 60mg of elementary iron are appropriate, as the not severe but disturbing gastrointestinal side effects are related to iron dosage and preparation. Consider enhancers (vitamin C) and prevent inhibitors (coffee, black tea, phytates, calcium) of iron uptake to increase absorption. Athletes and patients with repeatedly low ferritin values profit from an intermittent oral substitution to preserve iron stores, e.g. 2 iron tablets per week (containing 40 to 60mg elemental iron) as maintenance therapy. In cases of plausible non-responders, incompatibility with oral therapy, a concomitant disease (e.g. depression) or low iron stores before an altitude training camp, i.v. iron therapy should be considered. Long-term daily oral iron intake or iv-supplementation in the presence of normal or even high ferritin values is not recommended and may be harmful.
Résumé
La carence en fer est fréquente chez les sportifs. Tous les stades de la carence en fer ont un effet sur la performance et doivent être traités. Les mécanismes principaux qui en sont responsables chez les athlètes sont : besoins en fer élevés, augmentation des pertes de fer au niveau gastro-intestinal et un blocage de son absorption par l’élévation des taux d’hepcidine.
Les examens sanguins de base recommandés comprennent : hémoglobine, hématocrite, indices érythrocytaires, protéine C-réactive et ferritine.
Chez les athlètes en bonne santé des deux sexes, on admet qu’un taux de ferritine <15 µg/l correspond à des stocks de fer entièrement vides, et un taux entre 15 et 30 µg/l à des stocks réduits. On recommande ainsi une valeur de seuil à 30 µg/l pour le diagnostic de carence en fer. Chez des enfants de 6 à 12 ans et des adolescents de 12 à 15 ans, la valeur seuil recommandée se trouve à 15, respectivement 20 µg/l. On retient comme exception la situation des athlètes d’élite adultes se rendant en camp d’altitude, où il est pertinent de viser des valeurs à >50 µg/l, puisque les besoins en fer sont particulièrement élevés dans ce cas.
Le traitement de la carence en fer se base sur des conseils nutritionnels et une supplémentation per os ou, dans des cas exceptionnels, par voie intraveineuse. Les athlètes qui présentent des valeurs de ferritine basses à répétition bénéficient d’une complémentation orale intermittente pour maintenir les stocks de fer. Il est important de suivre les athlètes de manière individuelle par un contrôle sanguin de routine biannuel. Un traitement de fer per os à long terme ou un traitement par voie intraveineuse en présence de valeurs de ferritine normales ou élevées ne fait aucun sens et peut même être dommageable.
Acknowledgements
We thank all those who supported us to write this article. These include Prof. Dr. med. Arno Schmidt-Trucksäss,
Prof. Dr. med. Roland von Känel, Dr. med. Kerstin Warnke and Dr. med. Christian Schlegel.
Corresponding author
 German E. Clénin
German E. Clénin
Sportmedizinisches Zentrum
Ittigen bei Bern
Haus des Sports
3063 Ittigen, Switzerland
german.clenin@smzbi.ch
Literature
- Proposed nutrient and energy intakes for the European community: a report of the Scientific Committee for Food of the European community. Nutr. Rev. 51, 209–212 (1993).
- Egli, I. M. MARIA ANDERSSON, INES M. EGLI, MICHAEL B. ZIMMERMANN. at http://www.rosenfluh.ch/rosenfluh/articles/download/1318/Eisenmangel.pdf
- Schleiffenbaum, B. E. et al. Unexpected high prevalence of metabolic disorders and chronic disease among young male draftees – the Swiss Army XXI experience. Swiss Med Wkly 136, 175–184 (2006).
- Sandström, G., Börjesson, M. & Rödjer, S. Iron deficiency in adolescent female athletes – is iron status affected by regular sporting activity? Clin J Sport Med 22, 495–500 (2012).
- Latunde-Dada, G. O. Iron metabolism in athletes- achieving a gold standard. Eur. J. Haematol. (2012). doi:10.1111/ejh.12026
- Dubnov, G. et al. High prevalence of iron deficiency and anemia in female military recruits. Mil Med 171, 866–869 (2006).
- Cippa, P. & Krayenbühl, P.-A. Eisenmangel: Es geht nicht nur um Anämie. Schweiz Med Forum 11–12 (2014).
- Martius, F. Eisenmangel ohne Anämie – ein heisses Eisen. in Schweiz Med Forum 9, 294–9 (2009).
- Streuli, R. A. Ferrum bonum et laudabile (lucrosumque). in Schweiz Med Forum 8, 563 (2008).
- Colombani, P. C. & Mannhart, C. Energie- und Nährstoffaufnahme im Schweizer Spitzensport – eine erste Bestandsaufnahme zu Beginn des zweiten Jahrtausends. 7–16 (2003).
- Mettler, S. & Zimmermann, M. B. Iron excess in recreational marathon runners. Eur J Clin Nutr 64, 490–494 (2010).
- Ganz, T. Molecular Control of Iron Transport. JASN 18, 394–400 (2007).
- Löffler, G. in Biochemie mit Pathobiochemie. 7. Komplett überarbeitete Auflage. Springer Verlag. Heidelberg. S.416 ff (2008).
- Ganz, T. & Nemeth, E. Iron metabolism: interactions with normal and disordered erythropoiesis. Cold Spring Harb Perspect Med 2, a011668 (2012).
- Yip, R. in Present Knowledge in Nutrition, Bowman BA, Russell RM 311–328 (ILSI Press, Washington DC, 2001).
- Nemeth, E. & Ganz, T. Regulation of iron metabolism by hepcidin. Annu. Rev. Nutr. 26, 323–342 (2006).
- Young, B. & Zaritsky, J. Hepcidin for clinicians. Clin J Am Soc Nephrol 4, 1384–1387 (2009).
- Taylor, C. et al. Hematologic, iron-related, and acute-phase protein responses to sustained strenuous exercise. J. Appl. Physiol. 62,
464–469 (1987). - Peeling, P., Dawson, B., Goodman, C., Landers, G. & Trinder, D. Athletic induced iron deficiency: new insights into the role of inflammation, cytokines and hormones. Eur. J. Appl. Physiol. 103, 381–391 (2008).
- Peeling, P. Exercise as a mediator of hepcidin activity in athletes. Eur. J. Appl. Physiol. 110, 877–883 (2010).
- Ganz, T. Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation. Blood 102, 783–788 (2003).
- Newlin, M. K. et al. The effects of acute exercise bouts on hepcidin in women. Int J Sport Nutr Exerc Metab 22, 79–88 (2012).23. Gaudin, C., Zerath, E. & Guezennec, C. Y. Gastric lesions secondary to long-distance running. Dig. Dis. Sci. 35, 1239–1243 (1990).
- Peters, H. P., De Vries, W. R., Vanberge-Henegouwen, G. P. & Akkermans, L. M. Potential benefits and hazards of physical activity and exercise on the gastrointestinal tract. Gut 48, 435–439 (2001).
- Waller, M. F. & Haymes, E. M. The effects of heat and exercise on sweat iron loss. Med Sci Sports Exerc 28, 197–203 (1996).
- Jones, G. R. & Newhouse, I. Sport-related hematuria: a review. Clin J Sport Med 7, 119–125 (1997).
- Miller, B. J., Pate, R. R. & Burgess, W. Foot impact force and intravascular hemolysis during distance running. Int J Sports Med 9, 56–60 (1988).
- Telford, R. D. et al. Footstrike is the major cause of hemolysis during running. J. Appl. Physiol. 94, 38–42 (2003).
- Herklotz, R. & Huber, A. Labordiagnose von Eisenstoffwechselstörungen. in Schweiz Med Forum 10, 500–507 (2010).
- Brugnara, C., Laufer, M. R., Friedman, A. J., Bridges, K. & Platt, O. Reticulocyte hemoglobin content (CHr): early indicator of iron deficiency and response to therapy. Blood 83, 3100–3101 (1994).
- Thomas, C. & Thomas, L. Biochemical markers and hematologic indices in the diagnosis of functional iron deficiency. Clin. Chem. 48, 1066–1076 (2002).
- Macdougall, I. C. What is the most appropriate strategy to monitor functional iron deficiency in the dialysed patient on rhEPO therapy? Merits of percentage hypochromic red cells as a marker of functional iron deficiency. Nephrol. Dial. Transplant. 13, 847–849 (1998).
- D’ Onofrio, G., Zini, G., Ricerca, B. M., Mancini, S. & Mango, G. Automated measurement of red blood cell microcytosis and hypochromia in iron deficiency and beta-thalassemia trait. Arch. Pathol. Lab. Med. 116, 84–89 (1992).
- D’ Onofrio, G. et al. Simultaneous measurement of reticulocyte and red blood cell indices in healthy subjects and patients with microcytic and macrocytic anemia. Blood 85, 818–823 (1995).
- Brugnara, C., Zurakowski, D., DiCanzio, J., Boyd, T. & Platt, O. Reticulocyte hemoglobin content to diagnose iron deficiency in children. JAMA 281, 2225–2230 (1999).
- Franck, S., Linssen, J., Messinger, M. & Thomas, L. Potential utility of Ret-Y in the diagnosis of iron-restricted erythropoiesis. Clin. Chem. 50, 1240–1242 (2004).
- Schumacher, Y. O., Schmid, A., König, D. & Berg, A. Effects of exercise on soluble transferrin receptor and other variables of the iron status. Br J Sports Med 36, 195–199 (2002).
- Magge, H., Sprinz, P., Adams, W. G., Drainoni, M.-L. & Meyers, A. Zinc protoporphyrin and iron deficiency screening: trends and therapeutic response in an urban pediatric center. JAMA Pediatr 167, 361–367 (2013).
- Baart, A. M. et al. High prevalence of subclinical iron deficiency in whole blood donors not deferred for low hemoglobin. Transfusion 53, 1670–1677 (2013).
- Voss, S. C. et al. Variability of serum markers of erythropoiesis during 6 days of racing in highly trained cyclists. Int J Sports Med 35, 89–94 (2014).
- Cordova, A., Monserrat, J., Villa, G., Reyes, E. & Soto, M. A.-M. Effects of AM3 (Inmunoferon) on increased serum concentrations of interleukin-6 and tumour necrosis factor receptors I and II in cyclists. J Sports Sci 24, 565–573 (2006).
- Dickson, D. N., Wilkinson, R. L. & Noakes, T. D. Effects of ultra-marathon training and racing on hematologic parameters and serum ferritin levels in well-trained athletes. Int J Sports Med 3, 111–117 (1982).
- Fallon, K. E., Sivyer, G., Sivyer, K. & Dare, A. The biochemistry of runners in a 1600 km ultramarathon. Br J Sports Med 33, 264–269 (1999).
- Friedmann, B. Standards der Sportmedizin Sportleranämie. DEUTSCHE ZEITSCHRIFT FÜR SPORTMEDIZIN 52, (2001).
- Shaskey, D. J. & Green, G. A. Sports haematology. Sports Med 29, 27–38 (2000).
- Bärtsch, P., Mairbäurl, H. & Friedmann, B. [Pseudo-anemia caused by sports]. Ther Umsch 55, 251–255 (1998).
- Nichols, A. W. Nonorthopaedic problems in the aquatic athlete. Clin Sports Med 18, 395–411, viii (1999).
- Heinicke, K. et al. Blood volume and hemoglobin mass in elite athletes of different disciplines. Int J Sports Med 22, 504–512 (2001).
- Steiner, T. & Wehrlin, J. P. Does hemoglobin mass increase from age 16 to 21 and 28 in elite endurance athletes? Med Sci Sports Exerc 43, 1735–1743 (2011).
- Steiner, T. & Wehrlin, J. P. Comparability of haemoglobin mass measured with different carbon monoxide-based rebreathing procedures and calculations. Scand. J. Clin. Lab. Invest. 71, 19–29 (2011).
- Hinrichs, T. et al. Total hemoglobin mass, iron status, and endurance capacity in elite field hockey players. J Strength Cond Res 24, 629–638 (2010).
- Wehrlin, J. P. Live high-train low for 24 days increases hemoglobin mass and red cell volume in elite endurance athletes. Journal of Applied Physiology 100, 1938–1945 (2006).
- Garvican, L. A. et al. Intravenous Iron Supplementation in Distance Runners with Low or Suboptimal Ferritin. Med Sci Sports Exerc (2013). doi:10.1249/MSS.0b013e3182a53594
- Fellmann, N. Hormonal and plasma volume alterations following endurance exercise. A brief review. Sports Med 13, 37–49 (1992).
- Hamilton, T. et al. Die Radsport-Mafia und ihre schmutzigen Geschäfte: Der Insiderbericht über die Welt des Profiradsports: eine minutiöse Beichte, die erstmals das ganze … Armstrongs Schlüsselrolle darin aufzeigt. (2012).
- UCI La lutte contre le dopage: 40_ans.pdf. at http://oldsite.uci.ch/english/health_sante/docs/40_ans.pdf
- Bad blood : Nature News. at
- Athlete Biological Passport (ABP) Operating Guidelines, World Anti-Doping Agency. at https://www.wada-ama.org/en/resources/athlete-biological-passport/athlete-biological-passport-abp-operating-guidelines
- Saugy, M., Lundby, C. & Robinson, N. Monitoring of biological markers indicative of doping: the athlete biological passport. Br J Sports Med 48, 827–832 (2014).
- Davies, K. J., Maguire, J. J., Brooks, G. A., Dallman, P. R. & Packer, L. Muscle mitochondrial bioenergetics, oxygen supply, and work capacity during dietary iron deficiency and repletion. Am. J. Physiol. 242, E418–427 (1982).
- Davies, K. J. et al. Distinguishing effects of anemia and muscle iron deficiency on exercise bioenergetics in the rat. Am. J. Physiol. 246, E535–543 (1984).
- Finch, C. A. et al. Iron deficiency in the rat. Physiological and biochemical studies of muscle dysfunction. J. Clin. Invest. 58, 447–453 (1976).
- Burden, R. J. et al. Impact of Intravenous Iron on Aerobic Capacity and Iron Metabolism in Elite Athletes. Med Sci Sports Exerc (2014). doi:10.1249/MSS.0000000000000568.
- Garvican, L. A. et al. Intravenous iron supplementation in distance runners with low or suboptimal ferritin. Med Sci Sports Exerc 46, 376–385 (2014).
- DellaValle, D. M. & Haas, J. D. Iron supplementation improves energetic efficiency in iron-depleted female rowers. Med Sci Sports Exerc 46, 1204–1215 (2014).
- Waldvogel, S. et al. Clinical evaluation of iron treatment efficiency among non-anemic but iron-deficient female blood donors: a randomized controlled trial. BMC Med 10, 8 (2012).
- McClung, J. P. et al. Randomized, double-blind, placebo-controlled trial of iron supplementation in female soldiers during military training: effects on iron status, physical performance, and mood. Am. J. Clin. Nutr. 90, 124–131 (2009).
- Hinton, P. S. & Sinclair, L. M. Iron supplementation maintains ventilatory threshold and improves energetic efficiency in iron-deficient nonanemic athletes. Eur J Clin Nutr 61, 30–39 (2007).
- Brownlie, T., 4th, Utermohlen, V., Hinton, P. S. & Haas, J. D. Tissue iron deficiency without anemia impairs adaptation in endurance capacity after aerobic training in previously untrained women. Am. J. Clin. Nutr. 79, 437–443 (2004).
- Brownlie, T., 4th, Utermohlen, V., Hinton, P. S., Giordano, C. & Haas, J. D. Marginal iron deficiency without anemia impairs aerobic adaptation among previously untrained women. Am. J. Clin. Nutr. 75, 734–742 (2002).
- Friedmann, B., Weller, E., Mairbaurl, H. & Bärtsch, P. Effects of iron repletion on blood volume and performance capacity in young athletes. Med Sci Sports Exerc 33, 741–746 (2001).
- Hinton, P. S., Giordano, C., Brownlie, T. & Haas, J. D. Iron supplementation improves endurance after training in iron-depleted, nonanemic women. J. Appl. Physiol. 88, 1103–1111 (2000).
- WHO. WHO | Iron deficiency anaemia: assessment, prevention and control. WHO at http://www.who.int/nutrition/publications/micronutrients/anaemia_iron_deficiency/WHO_NHD_01.3/en/
- Hallberg, L. et al. Screening for iron deficiency: an analysis based on bone-marrow examinations and serum ferritin determinations in a population sample of women. Br. J. Haematol. 85, 787–798 (1993).
- Van Tellingen, O. The importance of drug-transporting P-glycoproteins in toxicology. Toxicol. Lett. 120, 31–41 (2001).
- Magnusson, B., Hallberg, L., Rossander, L. & Swolin, B. Iron metabolism and ’sports anemia’. I. A study of several iron parameters in elite runners with differences in iron status. Acta Med Scand 216, 149–155 (1984).
- Thomason, R. W. & Almiski, M. S. Evidence that stainable bone marrow iron following parenteral iron therapy does not correlate with serum iron studies and may not represent readily available storage iron. Am. J. Clin. Pathol. 131, 580–585 (2009).
- Krayenbuehl, P.-A., Battegay, E., Breymann, C., Furrer, J. & Schulthess, G. Intravenous iron for the treatment of fatigue in nonanemic, premenopausal women with low serum ferritin concentration. Blood 118, 3222–3227 (2011).
- Verdon, F. et al. Iron supplementation for unexplained fatigue in non-anaemic women: double blind randomised placebo controlled trial. BMJ 326, 1124 (2003).
- Vaucher, P., Druais, P.-L., Waldvogel, S. & Favrat, B. Effect of iron supplementation on fatigue in nonanemic menstruating women with low ferritin: a randomized controlled trial. CMAJ 184, 1247–1254 (2012).
- Gropper, S. S., Bader-Crowe, D. M., McAnulty, L. S., White, B. D. & Keith, R. E. Non-anemic iron depletion, oral iron supplementation and indices of copper status in college-aged females. J Am Coll Nutr 21, 545–552 (2002).
- Brutsaert, T. D. et al. Iron supplementation improves progressive fatigue resistance during dynamic knee extensor exercise in iron-depleted, nonanemic women. Am. J. Clin. Nutr. 77, 441–448 (2003).
- Clénin, G. E. Eisen im Sport – oft zu wenig, gelegentlich aber auch zu viel! Schweizerische Zeitschrift für Ernährungsmedizin 21–25 (2006).
- Mettler, S. Ferrum – ein Mineralstoff im Sport. SCHWEIZERISCHE ZEITSCHRIFT FUR SPORTMEDIZIN UND SPORTTRAUMATOLOGIE 52, 105–114 (2004).
- Mast, A. E., Blinder, M. A., Gronowski, A. M., Chumley, C. & Scott, M. G. Clinical utility of the soluble transferrin receptor and comparison with serum ferritin in several populations. Clin. Chem. 44, 45–51 (1998).
- Lamanca, J. J. & Haymes, E. M. Effects of low ferritin concentration on endurance performance. Int J Sport Nutr 2, 376–385 (1992).
- Yu, D., Huo, J., Xie, L. & Wang, L. [Meta-analysis of studies on cut-off value of serum ferritin for identifying iron deficiency]. Wei Sheng Yan Jiu 42, 228–235 (2013).
- Pitsis, G. C., Fallon, K. E., Fallon, S. K. & Fazakerley, R. Response of soluble transferrin receptor and iron-related parameters to iron supplementation in elite, iron-depleted, nonanemic female athletes. Clin J Sport Med 14, 300–304 (2004).
- Fallon, K. E. Screening for haematological and iron-related abnormalities in elite athletes-analysis of 576 cases. J Sci Med Sport 11,
329–336 (2008). - Fallon, K. E. Utility of hematological and iron-related screening in elite athletes. Clin J Sport Med 14, 145–152 (2004).
- Bothwell, T. H., Charlton, R. W., Cook, J. D. & Finch, C. A. IRon metabolism in man. Oxford, UK: Blackwell Scientific Publications (1979).
- Miret, S., Simpson, R. J. & McKie, A. T. Physiology and molecular biology of dietary iron absorption. Annu. Rev. Nutr. 23, 283–301 (2003).
- Zimmermann, M. B., Biebinger, R., Egli, I., Zeder, C. & Hurrell, R. F. Iron deficiency up-regulates iron absorption from ferrous sulphate but not ferric pyrophosphate and consequently food fortification with ferrous sulphate has relatively greater efficacy in iron-deficient individuals. Br. J. Nutr. 105, 1245–1250 (2011).
- Monsen, E. R. Iron nutrition and absorption: dietary factors which impact iron bioavailability. J Am Diet Assoc 88, 786–790 (1988).
- Hercberg, S., Preziosi, P. & Galan, P. Iron deficiency in Europe. Public Health Nutr 4, 537–545 (2001).
- Rimon, E. et al. Are we giving too much iron? Low-dose iron therapy is effective in octogenarians. Am. J. Med. 118, 1142–1147 (2005).
- Zimmermann, M. B. et al. Plasma hepcidin is a modest predictor of dietary iron bioavailability in humans, whereas oral iron loading, measured by stable-isotope appearance curves, increases plasma hepcidin. Am. J. Clin. Nutr. 90, 1280–1287 (2009).
- DellaValle, D. M. Iron supplementation for female athletes: effects on iron status and performance outcomes. Curr Sports Med Rep 12,
234–239 (2013). - Cancelo-Hidalgo, M. J. et al. Tolerability of different oral iron supplements: a systematic review. Curr Med Res Opin 29, 291–303 (2013).
- Arzneimittelkompendium, Ferinject ® / Unerwünschte Wirkungen at <https://www.compendium.ch.
- Ritzmann, P. Eisencarboxymaltose. pharma-kritik 32, 29–31 (2010).
- Rienso®, Lösung zur intravenösen Injektion (Ferumoxytol) – Swissmedic – at .
- Rienso®, Lösung zur intravenösen Injektion – Swissmedic – at https://www.swissmedic.ch/marktueberwachung/00135/00166/00707/index.html?lang=de.
- Arzneimittelinformation zu Venofer® (Fe-saccharosis) /Warnhinweise und Vorsichtsmassnahmen. at http://www.swissmedicinfo.ch/.
- Arzneimittelinformation zu Ferinject® / Warnhinweise und
Vorsichtsmassnahmen (Fe-carboxymaltosis). at . - Prohibited List. World Anti-Doping Agency at
- Demarmels Biasiutti, F. Die Regulation des Eisenstoffwechsels.
Schweiz Med Forum 630–632 (2009). - Koskenkorva-Frank, T. S., Weiss, G., Koppenol, W. H. & Burckhardt, S. The complex interplay of iron metabolism, reactive oxygen species, and reactive nitrogen species: insights into the potential of various iron therapies to induce oxidative and nitrosative stress. Free Radic. Biol. Med. 65, 1174–1194 (2013).
- Kohgo, Y., Ikuta, K., Ohtake, T., Torimoto, Y. & Kato, J. Body iron metabolism and pathophysiology of iron overload. Int J Hematol 88, 7–15 (2008).
- Steinboeck F, et al. The relevance of oxidative stress and cytotoxic DNA lesions for spontaneous mutagenesis in non-replicating yeast cells. Mutat Res.2010;688:47–52.
- Nowsheen S, et al. Accumulation of oxidatively induced clustered DNA lesions in human tumor tissues. Mutat Res. 2009;674:131–6
- Pingitore A, et al. Exercise and oxidative stress: Potential effects of antioxydant dietary strategies in sports. Nutrition.2015;31:916–22
- Powers SK, Nelson WB, Hudson MB. Exercise-induced oxidative stress in humans: cause and consequences. Free Radic Biol Med. 2011;51:942–50.
- Chua, A. C. G. et al. Dietary iron enhances colonic inflammation and IL-6/IL-11-Stat3 signaling promoting colonic tumor development in mice. PLoS ONE 8, e78850 (2013).
- Wurzelmann JI, Silver A, Schreinemachers DM, Sandler RS, Everson RB. Iron intake and the risk of colorectal cancer. Cancer Epidemiol Biomarkers Prev. 1996;5:503–7.
- Fonseca-Nunes, A., Jakszyn, P. & Agudo, A. Iron and cancer risk – a systematic review and meta-analysis of the epidemiological evidence. Cancer Epidemiol. Biomarkers Prev. 23, 12–31 (2014).
- Heath, A.-L. M. & Fairweather-Tait, S. J. Clinical implications of changes in the modern diet: iron intake, absorption and status. Best Pract Res Clin Haematol 15, 225–241 (2002).
- Rossander-Hulthen, L&Hallberg, L. in Iron Nutrition in Health and Disease (Hallberg L.&Asp N.G.) 149–156 (John Libbey&Co; London, UK, 1996).
- Constantini, N. W., Eliakim, A., Zigel, L., Yaaron, M. & Falk, B. Iron status of highly active adolescents: evidence of depleted iron stores in gymnasts. Int J Sport Nutr Exerc Metab 10, 62–70 (2000).
- Rowland, T. W. Iron deficiency in the young athlete. Pediatr. Clin. North Am. 37, 1153–1163 (1990).
- Spodaryk, K. Iron metabolism in boys involved in intensive physical training. Physiol. Behav. 75, 201–206 (2002).
- Fairweather-Tait, S. Iron requirements and prevalence of iron deficiency in adolescents. in Iron Nutrition in Health and Disease (Hallberg L.&Asp N.G.) 137–148 (John Libbey&Co; London, UK, 1996).
- Beard, J. L. Iron requirements in adolescent females. J. Nutr. 130, 440S–442S (2000).
- Hallberg, L. Iron requirements, iron balance and iron deficiency in menstruating and pregnant women. in Iron Nutrition in Health and Disease (Hallberg L.&Asp N.G.) 165–182 (John Libbey&Co; London, UK).
- Raunikar, R. A. & Sabio, H. Anemia in the adolescent athlete. Am. J. Dis. Child. 146, 1201–1205 (1992).
- Herklotz, R., Lüthi, I., Ottiger, C. & Huber, A. R. Referenzbereiche in der Hämatologie. Therapeutische Umschau 63, 5–24 (2006).
- Dietary Reference Intakes (DRIs): Estimated Average Requirements for Groups – 5_Summary Table Tables 1-4.pdf. at http://www.iom.edu/Activities/Nutrition/SummaryDRIs/~/media/Files/Activity%20Files/Nutrition/DRIs/5_Summary%20Table%20Tables%201-4.pdf







Comments are closed.