Tscholl P1, Meynard T1, Le Thanh N1, Neroladaki A2
1Service de chirurgie orthopédique et traumatologie de l’appareil locomoteur, Unité d’Orthopédie et de Traumatologie du Sport (UOTS), Swiss Olympic Medical Center – Cressy, Santé, Hôpitaux Universitaires de Genève, Genève, Suisse.
2Département de Radiologie, Hôpitaux Universitaires de Genève, Genève, Suisse.
Abstract
Muscle injuries are responsible for up to one third of the musculoskeletal injuries occuring in professional football and summer Olympic Games. Classifying injuries has the objective to define the extent, to predict the time-loss, and potentially to suggest a treatment plan to optimize the return to sport and prevent from re-injury.
Most recent classification systems have integrated patient’s history, clinical examination, and diagnostic imaging, to describe severity and therefore time-loss.
The different aspects and the most frequently used classification systems are discussed with special regard their validity to predict return to sport.
Résumé
Les lésions musculaires représentent jusqu’à un tiers des lésions musculo-squelettiques chez les joueurs professionnels de football et lors des jeux olympiques d’été. La classification de ces lésions a pour but de définir l’étendu des dommages, de prédire le temps de récupération et idéalement qui nous permettrait de cibler le traitement afin d’optimiser le retour à la pratique sportive et d’éviter la récidive de lésions musculaires, qui est l’une des principale complications.
Les classifications les plus récentes incluent les antécédents et comorbidités du patient, l’examen clinique ainsi que le bilan radiologique. Ces dernières sont principalement limitées dans la description de la sévérité des lésions ainsi que la prédiction du temps de récupération attendu.
Les systèmes de classifications les plus fréquemment utilisées ont été abordés, en particulier leur capacité à prédire le retour à la pratique sportive.
Introduction
Muscle injuries occur in up to 40 % of all professional football players per season[16] and are responsible for up to one third of all musculoskeletal injuries recorded during international football tournaments and Summer Olympic Games. [19] Most frequently, muscle injuries occur due to a sudden eccentric overload such as while sprinting, kicking, or due to an abrupt and forceful slippage, causing the indirect muscle injury. [1,25,33] Indirect muscle injuries are mainly found in muscles that bridge two joints (such as the rectus femoris of the quadriceps muscle, the medial gastrocnemius muscle or the hamstring muscle group) containing mainly fast-twitch type 2 fibers, which have greater contraction velocity and greater capacity to change length, however less capacity to withstand tension. [1,25,33] The location of injury usually is close to or at the myotendinous junction. Direct muscle injuries are found in less than 15 % of the cases and may occur in any muscle undergoing contusion. [49]
Ideally, a classification system permits to make a precise diagnosis and a prognosis for time-loss with the objective to allow to tailor an adequate treatment plan and finally to reduce the rate of reoccurrence injuries that have been described in up to 13 % of the cases. [49] Multiple classification systems have been published in literature, which are highlighted in this article.
Classification
Until the 1980s, the diagnosis and grading of the severity of a muscle lesion were solely based on patient’s history and physical exam. [34,40] With the development of imaging techniques such as ultrasound and MRI[45], knowledge of the muscular fascia and its connective tissue increased considerably in the context of muscle injuries, and therefore structural properties of the muscle damage and its precise location have been adopted to most of the muscle injury classification (see table 1). [10] Despite their classification being easily reproducible and objective, their reliability to predict return to sports has been shown of limited use. [56,57] Therefore, patient’s history, the clinical examination and the information on the morphological damage seen on MRI or US need to be included for the best understanding of a muscle injury. [16,31,38,51]
Injury mechanism and patient’s history
The observation of the injury mechanism in indirect muscle injuries, the immediate response by the athlete and its clinical examination are essential, especially to initiate adequate treatment strategies and to prevent further muscle damage. [21,59] Whereas the initial clinical exam might not be very contributive in terms of the extent of the muscle lesion and its required time of healing (except in the case of total loss of function and rapid swelling due to a hematoma for which complete muscle tear or tendinous avulsion is suspected and an imaging is absolutely required since surgical treatment might need to be considered), repeated exam between 48 to 72 hours might elicit with high precision the localization and the severity of the muscle injury. [31,56] A local hardening of muscular tissue will yet not tell, whether a structural injury has occurred or if there is only a functional (non-structural) muscle injury, however, combined with a significant loss of range of motion or a swelling, an injury to the aponeurotic tissue becomes probable, and the time for healing and return to sports will be increased. Athletes taking more than 1 day to walk without pain, greater pain at the time of injury (> 6 on the visual analogue pain scale), a popping sound at the time of injury, pain during every day activities for more than 3 days and greater than 15° motion-range limitation are associated with a loss of time more than 4 weeks. [21,59] The threshold size of a palpable gap might vary in literature from 2–5mm, however clearly indicates a structural muscle injury. [4,5,31,55]
Even though in most classification systems not considered, patient’s history as previous muscle injuries, underlying joint injuries, anterior pelvic tilt, a limitation in range of motion, eccentric strength deficit, fatigue and age are important to respect when tailoring a treatment plan and especially the return to sport to minimize recurrence. [2,13,14,31,36]


Diagnostic imaging
Ultrasound was the first imaging modality used to diagnose, grade and monitor the healing process of muscle injuries. The major advantages are its easy, quick and rather cheap access, enabling a dynamic view of the lesions, and proves to be as sensitive as MRI detecting the presence of an injury. [11] By means of ultrasound, puncture of the hematoma, which might be difficult to distinguish from normal muscle tissue within the first two days, is facilitated and is recommended by some authors, reducing pain and local inflammation. [32] MRI is known for being less observer-dependent imaging modality for sizing the injury and its retraction, and might show benefits for follow-up imaging to ultrasound. [11] Both techniques show poor prognostic value for return to sport. [11,37] The relevance of the size of the intramuscular edema, the amount of hematoma, and the intramuscular tendon involvement have so far not been found to have any predictive value for the time requested to return to play in hamstring injuries. [12,18,39,53,54] Furthermore, it is important to bear in mind, that only moderate muscle injuries can be made visible, and functional muscle injuries which account for 56 % of the days missed in professional football due to muscle lesions, will not be detected on imaging. [17]
Integrative classification systems
Currently, the most widely used and accepted classification published is the “Munich consensus statement” published by Mueller-Wohlfahrt et al.[31] which includes the injury mechanism (direct or indirect), early symptoms, signs on clinical exam, functional disorders with neuromuscular origin, and structural muscle lesions depending on their size and location (see table 2). They emphasize on the importance of functional muscle injuries and its different etiologies, which may be subtle and difficult to diagnose [23, 46], but most probably the underlying cause of recurrence or other overuse injuries. [15] They also distinguish between several etiologies of the muscle disorder and therefore highlight the multidisciplinary approach in treating muscle injuries. This classification system has been validated for thigh injuries in professional football players, and showed some predictive value in time-loss for non-structural and structural injuries however with a wide heterogeneity. [15] The classification systems by the British Athletics Medical team [38] and the Italian Society of Muscle, Ligament and Tendons (ISMuLT)[29] including anatomic localization of the injury (proximal, middle third or distal) are not currently used in scientific literature. The injury localization however is crucial. More proximal aponeurotic injuries to the rectus femoris were found to have greater lesion to the muscle fibers surrounding connective tissue and were associated with longer time-loss[6] and therefore might have to be classified as a higher grade. [31,51]
More recently, the MLG-R (faceted) classification system by Valle et al. [51] has been introduced for hamstring injuries, including not only the mechanism of injury (direct, indirect and MRI negative injuries) and the grading on MRI, but also its location of the muscle (proximal, middle third and distal) and the number of its re-occurrence. It represents the most complete and elaborated classification in literature especially in terms of its scientific value, however there is a lack of evidence that this approach will increase patient management and is practicable in clinical use. [23]
Clinical implication of classifying muscle injuries
Functional muscle injuries (type 1 and 2)
Each skeletal muscle fiber (muscle cell) contains a multitude of parallel aligned myofibrils that are attached to the sarcolemma (muscle cell membrane) by the desmin intermediate filament network, which permits to transmit the contractile force to the connective tissue and finally to the tendons (see figure 1). The smallest functional muscular unit – the sarcomere – is connected in series and is delimited on each side by z-discs, to which actin protein filaments are directly attached. The myosin protein filaments are attached to the z-discs by the titin protein. Eccentric work of the muscle fibers includes non-synchronized contraction of the myofilaments, which stretches the parallel aligned sarcomers and will injure the passive structures at the z-discs such as the titin filaments. Proteins such as filamine and BAG3 will be released from the z-discs, which induce autophagia and increase at the same time protein synthesis to regenerate muscle tissue of up to 3 % of the entire muscle mass per day. [50] This is part of the normal muscle regeneration process with a not clearly defined transition to type 1b lesions (DOMS).
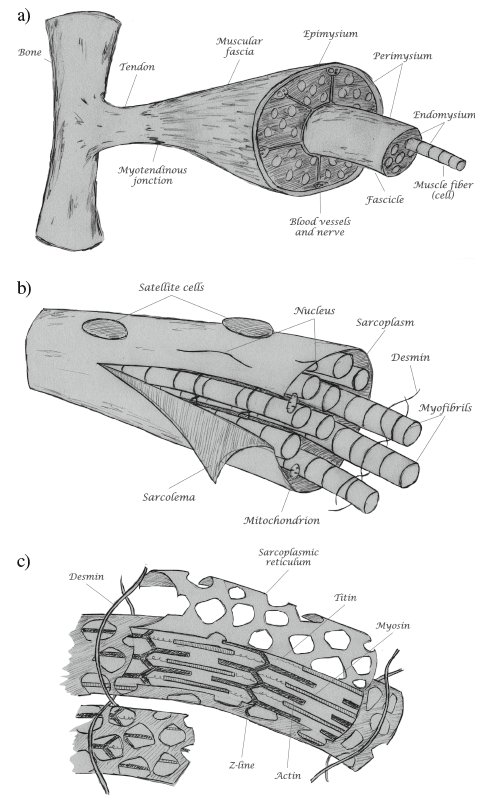
In these situations, vaso-active substances will be excreted which will increase vascular permeability, activate nociceptors and alter homeostasis of the muscle fiber, which can be seen on MRI as increased interstitial fluid (see figure 2). The intracellular process takes place mainly without cell disruption, whereby some inflammation by macrophages and neutrophil granulocytes may occur. [28] Clinically, an increase in muscle tone may be observed as in type 1a lesions and lead to dysfunction of the intramuscular coordination, and as a consequence increase considerably the risk for structural muscle damage. [35]
Muscle sprains corresponding to type 2a and 2b injuries, according to Müller-Wohlfahrt et al.[31], show a deregulation of the muscle tone, and appear typically suddenly and cramp-like. Athletes might feel to stretch or “to shake away their pain” which usually does not work, and might lead to worsening muscle pain. Continued exercise might lead to structural muscle injury. [35]




Minor to moderate muscle injuries (type 3)
The key component in structural muscle lesions is the connective tissue with its complex structure of the epi-, peri- and endomysium containing the neurovascular supply. It limits muscular bleeding and may partially prevent the muscle bundles from major retraction. Therefore, especially if the perimysium and the aponeurosis are injured, the time for healing is significantly increased. [31] Extracellular matrix will be degraded by proteases excreted by the tissue damage. Within the first 12–24 hours neutrophils will mobilize and stimulate macrophage cells for phagocytosis, which will be stimulating the satellite cells from day 1. [26] The maximum satellite cell activity (muscular precursor cells surrounding each muscle fiber) is found at the third day after the injury. These cells will fuse to myotubes or will fuse with pre-existing muscle fibers. Ideally, muscle fibers will be restored 14 days after the injury, however the size and also structural protein have not yet reached maturation. Interference with this macrophage induced process will lead to increased connective scar tissue formation and inclusion of fat. [44] It is key to minimize the destruction of the extracellular matrix by applying the PRICE protocol or even puncture the hematoma, ideally within 36hours after trauma. The use NSAIDs might be beneficial within the first 48–96 hours reducing the initial tissue damage, however should thereafter be limited due to interference with the satellite cell activity. [30, 47] Injection of Actovegin, Traumeel or PRP are suggested by some authors, such as different enzymes (Bromelain, Trypsin and Aescin), although current evidence is scarce. [8]
Type 3a injuries are of less than 5mm size, hence include 1–5 primary muscle bundles (being 1mm2 each). The injury to the perimysium is only minor, whereas the aponeurosis might be still intact. Type 3a injuries usually heal without defect (see figure 3a). However, injury to the perimysium and the aponeurosis is found in type 3b injuries (> 5 mm), which is the cause for intermuscular hematoma formation (see figure 3b). Treatment and lay-off time is very heterogeneous and might last from 10 to 40 days depending on especially the injury to the connective tissue and even more the aponeurosis (for type 3a injuries). Clinical examination 48 to 72 hours after injury is very helpful to early differentiate between a type 3a or type 3b injury, since early active mobilization must be initiated with minor injuries to improve the muscle regeneration process, and not before day 7 in type 3b injuries. [42] Massage of the injured site should be avoided, since it might favor the formation of myositis ossificans. Too early return to sport might lead especially in higher grade muscle injuries to over stimulation of the extracellular matrix and therefore stiffening of the muscle tissue at the site of injury.
Complete muscle disruption and tendon avulsions (type 4)
Complete muscle disruption is rare, and mostly implies tendon avulsion injuries, especially of the ischiocrural muscles or the rectus femoris muscle (see figure 4). Hematoma formation will be important (except in chronic tendinopathies) and will require early PRICE treatment. Proximal tendon avulsion at the sciatic tubercle can be treated conservatively in most of the cases, although surgical treatment is recommended for a two-tendon injury in professional athletes or high demand patients. [27]
Direct muscle injuries (type 5)
The grade and size of direct muscle injuries depend on the intensity, the velocity and the state of contraction at the time of contact, and can occur at almost any localization of the muscle. It is most found intra-muscularly, and only rarely at the myotendinus junction, contrary to the indirect muscle lesions. An increase of the intracompartimental pressure may occur and lead to an acute compartment syndrome which should never be missed. In general, direct muscle injuries are of lesser degree than indirect muscle injuries, since there is no loss of continuity of the muscle fibers, and therefore the mean lay-off time is expected between 2–3 weeks.[49] Protecting further muscle damage by applying the PRICE protocol, immediate immobilizing the quadriceps muscle for example in 120° of knee flexion has shown favorable results. [3] Myositis ossificans can develop especially from a contusion to the vastus intermedius muscle. The risk can be diminished by avoiding too early return to sport and deep friction massages. [7]



Criteria for predicting time-loss,
return to sport and recurrent injury
Longer time to recovery is found in indirect muscle injuries, greater deficit in range of motion, pain while walking 24–72 hours after the injury, time to first consultation > 1 week with a delay of active rehabilitation, proximal tendon involvement, shorter distance to the ischial tuberosity and the antero-inferior iliac spine [20], whereas other signs on US or MR imaging show only weak predicting power. [12,18,39,53,54]
Re-injuries most frequently occur early after return to sport, and at the same localization with a greater extent. [58] The biceps femoris muscle seems to be more prone than the semitendinosus or semimembranosus muscle. [54]
Whereas rehabilitation protocols in literature rely on individual’s perception on pain and apprehension [24], return to sport criteria are mostly relying on clinical assessment and performance tests. [52] Persisting intra-muscular signal intensity might still be visible in 89 % of the patients after they have clinically fully recovered, making MRI not a valid tool for the decision-making process of return to play. [41] The most frequently mentioned return to play criteria in literature are pain-free palpation, symmetrical range of motion, similar strength at concentric contraction of 5–20 % difference, a Hamstring-Quadriceps ratio of > 0.55, athletes self-perceiving, no difference in single-leg triple-hop test and similar competing levels as before. [52] Important to mention, that studies relying on dynamometric isokinetic testing having the earliest return to play, whereas studies relying on clinical assessment such as the Askling H-test and patients performing a rehabilitation protocol emphasizing on eccentric hamstring exercises had the lowest rate of re-injury (0–3.6 %). [24,48]
Conclusion
There is no classification system for muscle injuries in literature that comprehends all major factors describing the severity and especially the estimated time-loss until return to sport. Imaging is of clinical importance especially in higher grade structural muscle injuries, where surgical treatment must be evaluated. However, early detection of patient related triggers and risk factors, repetitive clinical follow-up exam, combined with the knowledge of the muscle healing process are crucial elements to establish an adequate treatment plan in muscle injuries.

Corresponding author
Dr. med. Philippe M. Tscholl
Rue Gabrielle Perret-Gentil 4
1205 Genève, Suisse.
Telephone: +41 22 727 15 50
Philippe.Tscholl@hcuge.ch
References
- Anderson K, Strickland SM, Warren R (2001) Hip and groin injuries in athletes. Am J Sports Med 29:521-533.
- Arnason A, Andersen TE, Holme I, Engebretsen L, Bahr R (2008) Prevention of hamstring strains in elite soccer: an intervention study. Scand J Med Sci Sports 18:40-48.
- Aronen JG, Garrick JG, Chronister RD, McDevitt ER (2006) Quadriceps contusions: clinical results of immediate immobilization in 120 degrees of knee flexion. Clin J Sport Med 16:383-387.
- Askling CM, Tengvar M, Saartok T, Thorstensson A (2007) Acute first-time hamstring strains during high-speed running: a longitudinal study including clinical and magnetic resonance imaging findings. Am J Sports Med 35:197-206.
- Askling CM, Tengvar M, Saartok T, Thorstensson A (2007) Acute first-time hamstring strains during slow-speed stretching: clinical, magnetic resonance imaging, and recovery characteristics. Am J Sports Med 35:1716-1724.
- Balius R, Maestro A, Pedret C, Estruch A, Mota J, Rodriguez L, et al. (2009) Central aponeurosis tears of the rectus femoris: practical sonographic prognosis. Br J Sports Med 43:818-824.
- Beiner JM, Jokl P (2002) Muscle contusion injury and myositis ossificans traumatica. Clin Orthop Relat Res S110-119.
- Bloch W, Hotfiel T, Ueblacker P, Tischer T, Bily W, Engelhardt M. Anatomie/Physiologie der Muskelheilung. In: Engelhardt M, Mauch R, eds. Muskel- und Sehneverletzungen. Jena: Vopelius; 2017:59-73.
- Bryan Dixon J (2009) Gastrocnemius vs. soleus strain: how to differentiate and deal with calf muscle injuries. Curr Rev Musculoskelet Med 2:74-77.
- Chan O, Del Buono A, Best TM, Maffulli N (2012) Acute muscle strain injuries: a proposed new classification system. Knee Surg Sprots Traumatol Arthrosc 20:2356-2362.
- Connell DA, Schneider-Kolsky ME, Hoving JL, Malara F, Buchbin-
der R, Koulouris G, et al. (2004) Longitudinal study comparing sonographic and MRI assessments of acute and healing hamstring injuries. AJR Am J Roentgenol 183:975-984. - Crema MD, Godoy IRB, Abdalla RJ, de Aquino JS, Ingham SJM, Skaf AY (2018) Hamstring Injuries in Professional Soccer Players: Extent of MRI-Detected Edema and the Time to Return to Play. Sports Health 10:75-79.
- Daly C, Persson UM, Twycross-Lewis R, Woledge RC, Morrissey D (2016) The biomechanics of running in athletes with previous hamstring injury: A case-control study. Scand J Med Sci Sports 26:413-420
- de Visser HM, Reijman M, Heijboer MP, Bos PK (2012) Risk factors of recurrent hamstring injuries: a systematic review. Br J Sports Med 46:124-130.
- Ekstrand J, Askling C, Magnusson H, Mithoefer K (2013) Return to play after thigh muscle injury in elite football players: implementation and validation of the Munich muscle injury classification. Br J Sports Med 47:769-774.
- Ekstrand J, Hagglund M, Walden M (2011) Epidemiology of muscle injuries in professional football (soccer). Am J Sports Med 39:1226-1232.
- Ekstrand J, Healy JC, Walden M, Lee JC, English B, Hagglund M (2012) Hamstring muscle injuries in professional football: the correlation of MRI findings with return to play. Br J Sports Med 46:112-117.
- Ekstrand J, Lee JC, Healy JC (2016) MRI findings and return to play in football: a prospective analysis of 255 hamstring injuries in the UEFA Elite Club Injury Study. Br J Sports Med 50:738-743.
- Engebretsen L, Soligard T, Steffen K, Alonso JM, Aubry M, Budgett R, et al. (2013) Sports injuries and illnesses during the London Summer Olympic Games 2012. Br J Sports Med 47:407-414.
- Fournier-Farley C, Lamontagne M, Gendron P, Gagnon DH (2016) Determinants of Return to Play After the Nonoperative Management of Hamstring Injuries in Athletes: A Systematic Review. Am J Sports Med 44:2166-2172.
- Guillodo Y, Here-Dorignac C, Thoribe B, Madouas G, Dauty M, Tassery F, et al. (2014) Clinical predictors of time to return to competition following hamstring injuries. Muscles Ligaments Tendons J 4:386-390.
- Gyftopoulos S, Rosenberg ZS, Schweitzer ME, Bordalo-Rodrigues M (2008) Normal anatomy and strains of the deep musculotendinous junction of the proximal rectus femoris: MRI features. AJR Am J Roentgenol 190:W182-186.
- Hamilton B, Alonso JM, Best TM (2017) Time for a paradigm shift in the classification of muscle injuries. J Sport Health Sci 6:255-261.
- Hickey JT, Timmins RG, Maniar N, Williams MD, Opar DA (2017) Criteria for Progressing Rehabilitation and Determining Return-to-Play Clearance Following Hamstring Strain Injury: A Systematic Review. Sports Med 47:1375-1387.
- Jarvinen TA, Jarvinen TL, Kaariainen M, Kalimo H, Jarvinen M (2005) Muscle injuries: biology and treatment. Am J Sports Med 33:745-764.
- Kharraz Y, Guerra J, Mann CJ, Serrano AL, Munoz-Canoves P (2013) Macrophage plasticity and the role of inflammation in skeletal muscle repair. Mediators Inflamm 2013:491497.
- Lempainen L, Banke IJ, Johansson K, Brucker PU, Sarimo J, Orava S, et al. (2015) Clinical principles in the management of hamstring injuries. Knee Surg Sports Traumatol Arthrosc 23:2449-2456.
- Lewis PB, Ruby D, Bush-Joseph CA (2012) Muscle soreness and delayed-onset muscle soreness. Clin Sports Med 31:255-262.
- Maffulli N, Oliva F, Frizziero A, Nanni G, Barazzuol M, Via AG, et al. (2013) ISMuLT Guidelines for muscle injuries. Muscles Ligaments Tendons J 3:241-249.
- Mikkelsen UR, Langberg H, Helmark IC, Skovgaard D, Andersen LL, Kjaer M, et al. (2009) Local NSAID infusion inhibits satellite cell proliferation in human skeletal muscle after eccentric exercise. J Appl Physiol (1985) 107:1600-1611.
- Mueller-Wohlfahrt HW, Haensel L, Mithoefer K, Ekstrand J, English B, McNally S, et al. (2013) Terminology and classification of muscle injuries in sport: the Munich consensus statement. Br J Sports Med 47:342-350.
- Mueller-Wohlfahrt HW, Ueblacker P, Haensel L, Garrett WE, Jr. Muscle Injuries in Sports. Stuttgart, New York: Georg Thieme Verlag; 2013.
- Noonan TJ, Garrett WE, Jr. (1999) Muscle strain injury: diagnosis and treatment. J Am Acad Orthop Surg 7:262-269.
- O’Donoghue DH. Treatment of injuries to athletes. 1st ed. Philadelphia: Saunders Company; 1962.
- Opar DA, Williams MD, Shield AJ (2012) Hamstring strain injuries: factors that lead to injury and re-injury. Sports Med 42:209-226.
- Orchard J, Best TM, Verrall GM (2005) Return to play following muscle strains. Clin J Sport Med 15:436-441.
- Petersen J, Thorborg K, Nielsen MB, Skjodt T, Bolvig L, Bang N, et al. (2014) The diagnostic and prognostic value of ultrasonography in soccer players with acute hamstring injuries. Am J Sports Med 42:399-404.
- Pollock N, James SL, Lee JC, Chakraverty R (2014) British athletics muscle injury classification: a new grading system. Br J Sports Med 48:1347-1351.
- Prakash A, Entwisle T, Schneider M, Brukner P, Connell D (2017) Connective tissue injury in calf muscle tears and return to play: MRI correlation. Br J Sports Med;10.1136/bjsports-2017-098362.
- Rachun A. Standard nomenclature of athletic injuries. 1st ed. Chicago, IL: American Medical Association; 1966.
- Reurink G, Goudswaard GJ, Tol JL, Almusa E, Moen MH, Weir A, et al. (2014) MRI observations at return to play of clinically recovered hamstring injuries. Br J Sports Med 48:1370-1376.
- Richard-Bulteau H, Serrurier B, Crassous B, Banzet S, Peinnequin A, Bigard X, et al. (2008) Recovery of skeletal muscle mass after extensive injury: positive effects of increased contractile activity. Am J Physiol Cell Physiol 294:C467-476.
- Schneider-Kolsky ME, Hoving JL, Warren P, Connell DA (2006) A comparison between clinical assessment and magnetic resonance imaging of acute hamstring injuries. Am J Sports Med 34:1008-1015.
- Sciorati C, Rigamonti E, Manfredi AA, Rovere-Querini P (2016) Cell death, clearance and immunity in the skeletal muscle. Cell Death Differ 23:927-937.
- Takebayashi S, Takasawa H, Banzai Y, Miki H, Sasaki R, Itoh Y, et al. (1995) Sonographic findings in muscle strain injury: clinical and MR imaging correlation. J Ultrasound Med 14:899-905.
- Tol JL, Hamilton B, Best TM (2013) Palpating muscles, massaging the evidence? An editorial relating to ‘Terminology and classification of muscle injuries in sport: The Munich consensus statement’. Br J Sports Med 47:340-341.
- Tscholl PM, Gard S, Schindler M (2016) A sensible approach to the use of NSAIDs in sports medicine. Swiss Sports Exerc Med 65:15-20.
- Tyler TF, Schmitt BM, Nicholas SJ, McHugh MP (2017) Rehabilitation After Hamstring-Strain Injury Emphasizing Eccentric Strengthening at Long Muscle Lengths: Results of Long-Term Follow-Up. J Sport Rehabil 26:131-140.
- Ueblacker P, Muller-Wohlfahrt HW, Ekstrand J (2015) Epidemiological and clinical outcome comparison of indirect (‘strain’) versus direct (“contusion”) anterior and posterior thigh muscle injuries in male elite football players: UEFA Elite League study of 2287 thigh injuries (2001-2013). Br J Sports Med 49:1461-1465.
- Ulbricht A, Gehlert S, Leciejewski B, Schiffer T, Bloch W, Hohfeld J (2015) Induction and adaptation of chaperone-assisted selective autophagy CASA in response to resistance exercise in human skeletal muscle. Autophagy 11:538-546.
- Valle X, Alentorn-Geli E, Tol JL, Hamilton B, Garrett WE, Jr., Pruna R, et al. (2017) Muscle Injuries in Sports: A New Evidence-Informed and Expert Consensus-Based Classification with Clinical Application. Sports Med 47:1241-1253.
- van der Horst N, van de Hoef S, Reurink G, Huisstede B, Backx F (2016) Return to Play After Hamstring Injuries: A Qualitative Systematic Review of Definitions and Criteria. Sports Med 46:899-912.
- van der Made AD, Almusa E, Whiteley R, Hamilton B, Eirale C, van Hellemondt F, et al. (2018) Intramuscular tendon involvement on MRI has limited value for predicting time to return to play following acute hamstring injury. Br J Sports Med 52:83-88.
- van Heumen M, Tol JL, de Vos RJ, Moen MH, Weir A, Orchard J, et al. (2017) The prognostic value of MRI in determining reinjury risk following acute hamstring injury: a systematic review. Br J Sports Med 51:1355-1363.
- Verrall GM, Slavotinek JP, Barnes PG, Fon GT (2003) Diagnostic and prognostic value of clinical findings in 83 athletes with posterior thigh injury: comparison of clinical findings with magnetic resonance imaging documentation of hamstring muscle strain. Am J Sports Med 31:969-973.
- Wangensteen A, Almusa E, Boukarroum S, Farooq A, Hamilton B, Whiteley R, et al. (2015) MRI does not add value over and above patient history and clinical examination in predicting time to sport after acute hamstring injuries: a prosepective cohort of 180 male athletes. Br J Sports Med 49:1579-1587.
- Wangensteen A, Guermazi A, Tol JL, Roemer FW, Hamilton B, Alonso JM, et al. (2018) New MRI muscle classification sytems and associations with return to sport after acute hamstring injuries: a prospective study. Eur Radiol;10.1007/s00330-017-5125-0.
- Wangensteen A, Tol JL, Witvrouw E, Van Linschoten R, Almusa E, Hamilton B, et al. (2016) Hamstring Reinjuries Occur at the Same Location and Early After Return to Sport: A Descriptive Study of MRI-Confirmed Reinjuries. Am J Sports Med 44:2112-2121.
- Warren P, Gabbe BJ, Schneider-Kolsky M, Bennell KL (2010) Clinical predictors of time to return to competition and of recurrence following hamstring strain in elite Australian footballers. Br J Sports Med 44:415-419.







Comments are closed.